

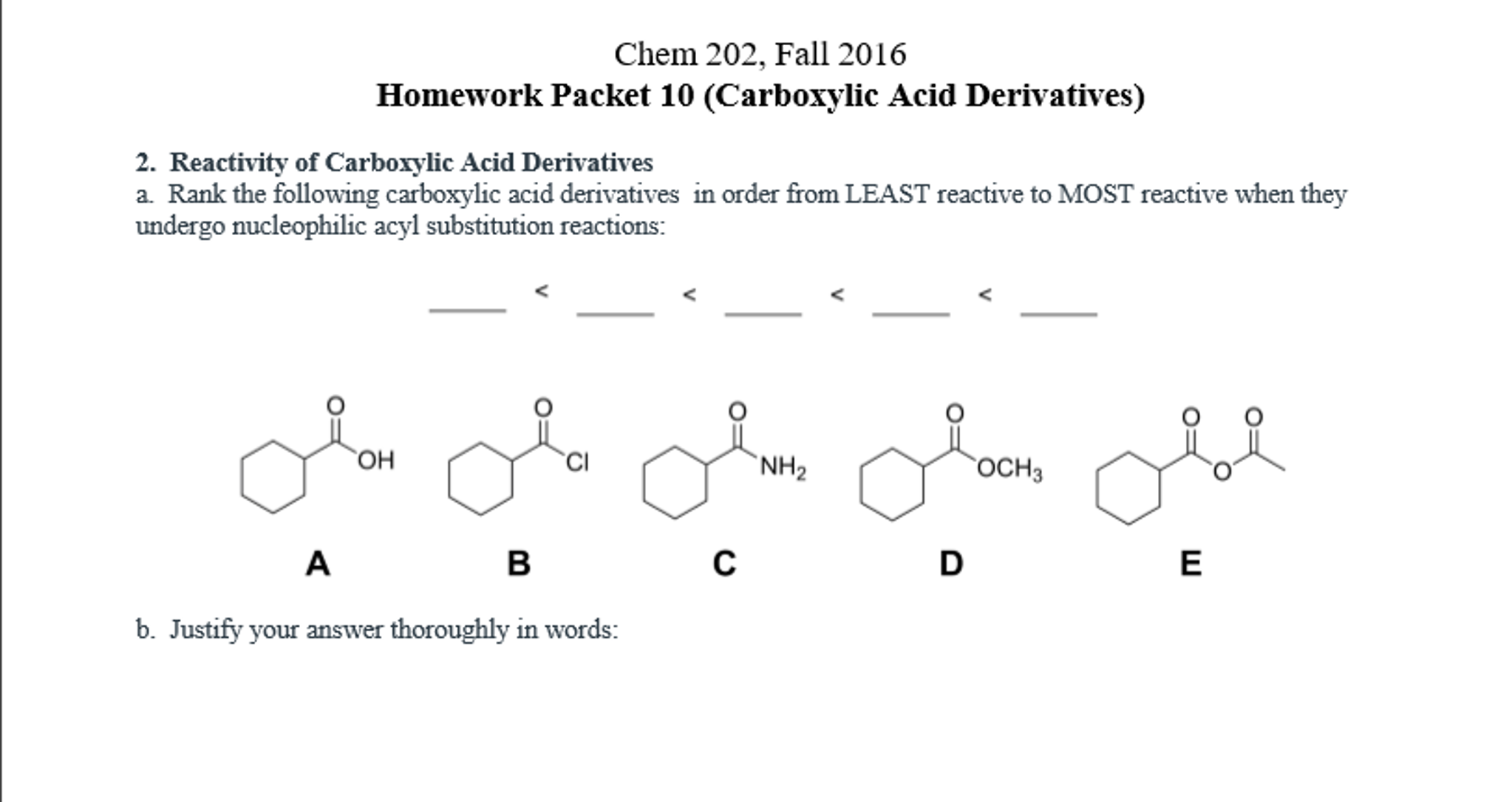
electronic effects: groups that can redistribute and stabilize negative charges are good leaving groups.steric effects: bulky groups around the C=O group helps protect the carbon center from nucleophilic attack.Proteins are made of peptide bonds, and they are very stable. Also, the C-N bond has a partial double bond characteristic. Amides are the most stable derivatives because NR 2 - is a terrible leaving group.Acid halides are the most reactive derivatives because halides are very good leaving groups.relative reactivity of acid derivatives: Acid chloride > Anhydride > Esters > Amides.hydrolysis of amides: the leaving group is not NR 2 -, it is the neutral amine.hydrolysis of fats and glycerides (saponification): saponification is basically the hydrolysis of an ester in base.transesterification: Ester + alcohol → new ester.See figure below for detailed mechanism of the Hofmann degradation and how the aryl group migrates. The alkyl migration is basically how the -R group on the other side of the C=O migrates and attaches itself to the nitrogen atom. Hofmann rearrangement: Hofmann rearrangement takes away the C=O of an amide.nucleophilic substitution: Nucleophile attacks the carbon center of the C=O group.Acid chloride + water → Carboxylic acid.Acid chloride + alcohol + base → Ester.Acid chloride + carboxylic acid + base → Anhydride.

Carboxylic acid + carboxylic acid + heat → Anhydride.Carboxylic acid + SOCl 2 → Acid chloride.The C-O ether stretch shows up around 1200 cm -1 Important reactions Ester: C=O group shows up at 1700 cm -1.Amide: the N-H shows up around 3300 cm -1, the C=O shows up at 1700 cm -1.Instead, 2 bands shows up between 1700 cm - cm -1. Anhydride: the double C=O doesn't show up as a single band.Acid chloride: the C=O will show up at greater than 1700 cm -1, pretty close to 1800 cm -1.


 0 kommentar(er)
0 kommentar(er)
